Compound details
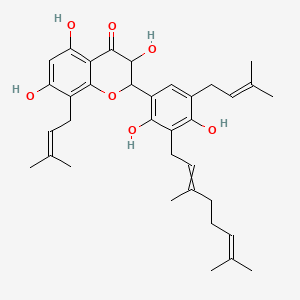
Sanggenol E
| Compound ID | CDAMM02864 |
|---|---|
| Common name | Sanggenol E | IUPAC name | 2-[3-(3,7-dimethylocta-2,6-dienyl)-2,4-dihydroxy-5-(3-methylbut-2-enyl)phenyl]-3,5,7-trihydroxy-8-(3-methylbut-2-enyl)-2,3-dihydrochromen-4-one |
| Molecular formula | C35H44O7 |
Experimental data
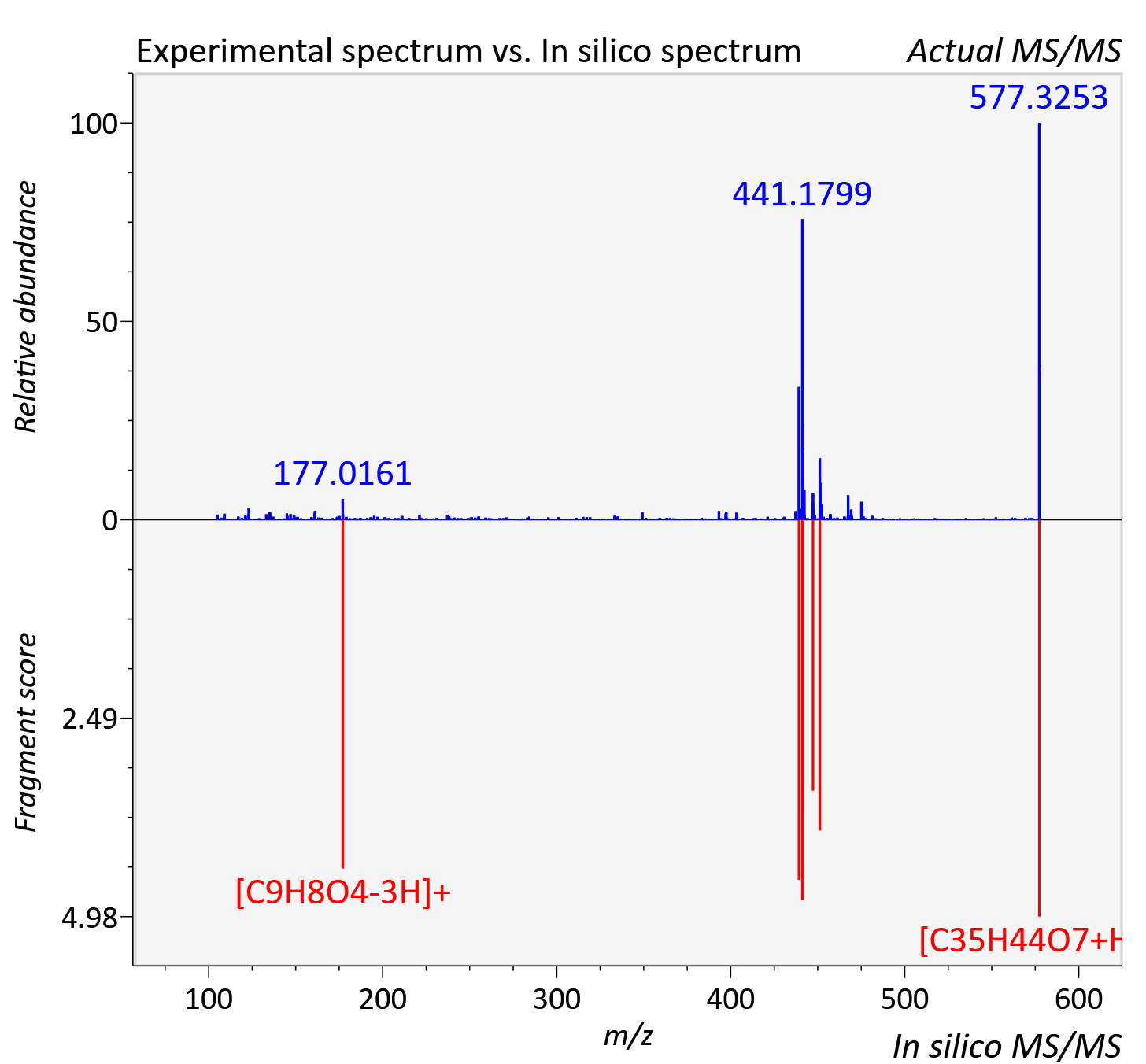
| Retention time | 15.55 |
|---|---|
| Adduct | [M+H]+ |
| Actual mz | 577.318 | Theoretical mz | 577.316 |
| Error | 3.68 |
| Ionizaton mode | Positive |
| Instrument type | LC-MS/MS-QTOF, spectrum predicted by MS-DIAL v4.9 integrated with MS-FINDER 3.60 |
| Score | 6.5542 |
Identifiers and class information
| Inchi key | ACDWEIMFJUNXIU-XXHIOHRXNA-N |
|---|---|
| Smiles | O=C1C2=C(O)C=C(O)C(=C2OC(C3=CC(=C(O)C(=C3O)CC=C(C)CCC=C(C)C)CC=C(C)C)C1O)CC=C(C)C |
| Superclass | Phenylpropanoids and polyketides |
| Class | Flavonoids |
Plant source
Pharmacokinetic properties
Compound-target network
Protein targets associated with phytocompound
| Uniprot ID | Gene name | Target name | TTD_ID | Prediction source |
|---|---|---|---|---|
| P22303 | ACHE | Acetylcholinesterase | T30082 | SwissTargetPrediction |
| P11926 | ODC1 | Ornithine decarboxylase | T60366 | SEA |
| P18031 | PTPN1 | Protein-tyrosine phosphatase 1B | T16347 | SEA |
| P19793 | RXRA | Retinoid X receptor alpha (by homology) | T13726 | SwissTargetPrediction |
| P30307 | CDC25C | Dual specificity phosphatase Cdc25C | T40569 | SEA |
| O60911 | CTSV | Cathepsin (V and K) | T93653 | SEA |
Target associated diseases
| TTD_ID | Disease_ID | Disease name | ICD_11 | Uniprot ID | Gene names |
|---|---|---|---|---|---|
| T30082 | DI0025 | Alzheimer disease | [ICD-11: 8A20] | P22303 | ACHE |
| T30082 | DI0166 | Glaucoma | [ICD-11: 9C61] | P22303 | ACHE |
| T30082 | DI0282 | Myasthenia gravis | [ICD-11: 8C6Y] | P22303 | ACHE |
| T30082 | DI0313 | Oesophageal/gastroduodenal disorder | [ICD-11: DD90] | P22303 | ACHE |
| T30082 | DI0332 | Pediculosis | [ICD-11: 1G00] | P22303 | ACHE |
| T30082 | DI0421 | Unspecific substance harmful effect | [ICD-11: NE6Z] | P22303 | ACHE |
| T60366 | DI0020 | African trypanosomiasis | [ICD-11: 1F51] | P11926 | ODC1 |
| T16347 | DI0009 | Acute diabete complication | [ICD-11: 5A2Y] | P18031 | PTPN1 |
| T16347 | DI0062 | Breast cancer | [ICD-11: 2C60-2C6Y] | P18031 | PTPN1 |
| T16347 | DI0308 | Obesity | [ICD-11: 5B80-5B81] | P18031 | PTPN1 |
| T16347 | DI0417 | Type 2 diabetes mellitus | [ICD-11: 5A11] | P18031 | PTPN1 |
| T13726 | DI0225 | Kaposi sarcoma | [ICD-11: 2B57] | P19793 | RXRA |
| T13726 | DI0283 | Mycosis fungoides | [ICD-11: 2B01] | P19793 | RXRA |
| T93653 | DI0055 | Bone cancer | [ICD-11: 2B5Z] | O60911 | CTSV |
| T93653 | DI0087 | Chronic pain | [ICD-11: MG30] | O60911 | CTSV |
