Compound details
C-curarine
| Compound ID | CDAMM02391 |
|---|---|
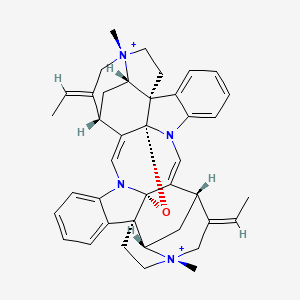
| Common name | C-curarine | IUPAC name | (1R,9R,11R,13S,15S,16S,19R,29S,30E,32S,35S,38E)-30,38-di(ethylidene)-16,32-dimethyl-10-oxa-8,26-diaza-16,32-diazoniadodecacyclo[27.5.2.213,16.18,12.01,9.02,7.09,28.011,19.011,26.015,19.020,25.032,35]nonatriaconta-2,4,6,12(39),20,22,24,27-octaene |
| Molecular formula | C40H44N4O |
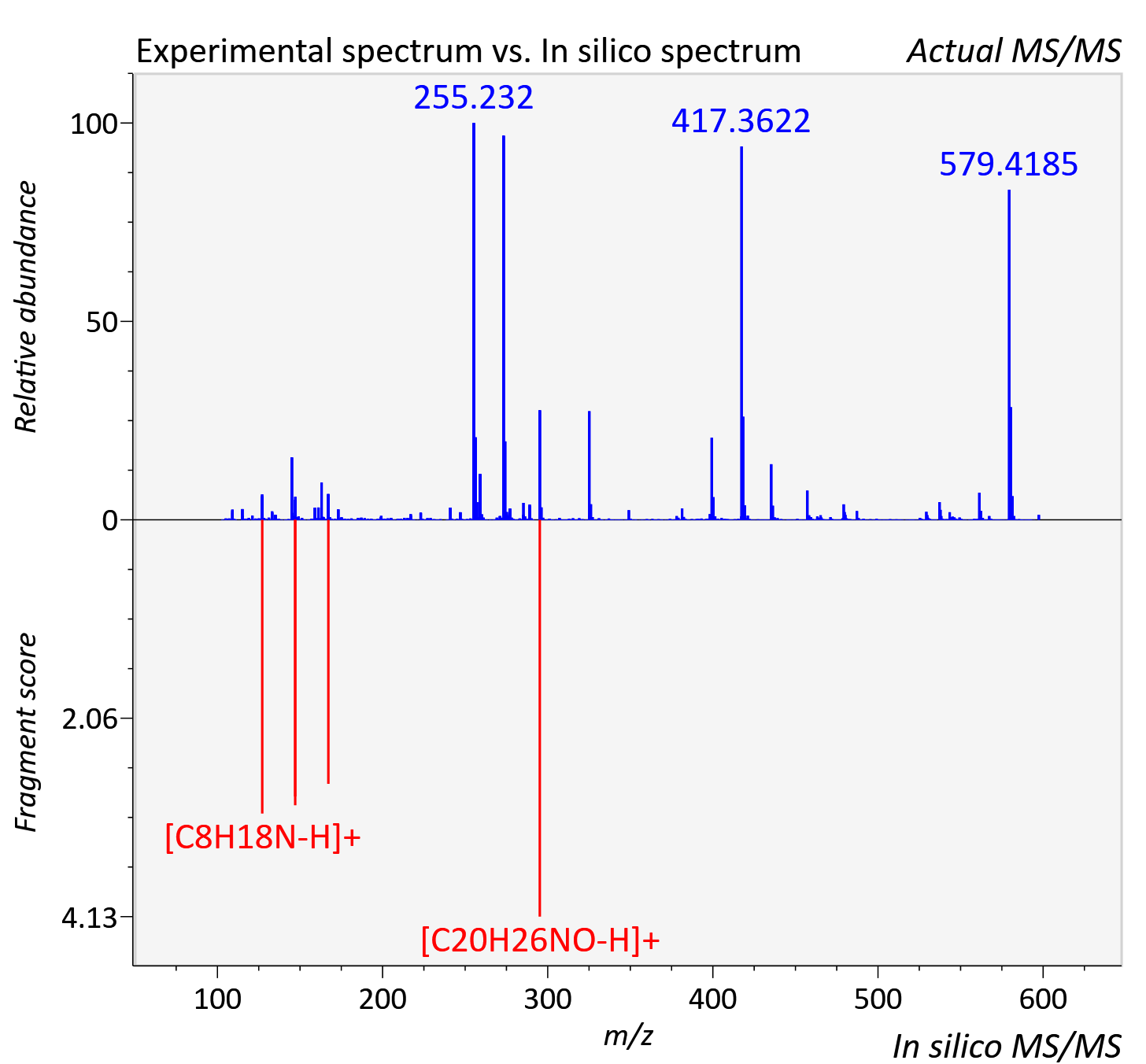
Experimental data
| Retention time | 4.5 |
|---|---|
| Adduct | [M+H]+ |
| Actual mz | 597.359 | Theoretical mz | 597.359 |
| Error | 0.46 |
| Ionizaton mode | Positive |
| Instrument type | LC-MS/MS-QTOF, spectrum predicted by MS-DIAL v4.9 integrated with MS-FINDER 3.60 |
| Score | 5.1921 |
Identifiers and class information
| Inchi key | DWELRYDMYVJVSL-ZDAJIBGCNA-N |
|---|---|
| Smiles | O1C23C4=CN5C=6C=CC=CC6C78CC[N+]9(C)CC(=CC)C(C(=CN3C=%10C=CC=CC%10C%112CC[N+]%12(C)CC(=CC)C4CC%11%12)C157)CC89 |
| Superclass | Alkaloids and derivatives |
| Class | Strychnos alkaloids |
Plant source
Pharmacokinetic properties
Compound-target network
Protein targets associated with phytocompound
| Uniprot ID | Gene name | Target name | TTD_ID | Prediction source |
|---|
Target associated diseases
| TTD_ID | Disease_ID | Disease name | ICD_11 | Uniprot ID | Gene names |
|---|
