Compound details
Calenduloside H
| Compound ID | CDAMM02182 |
|---|---|
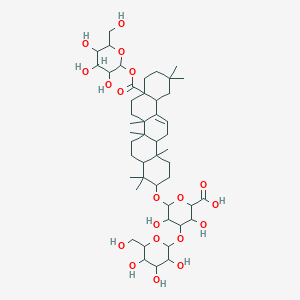
| Common name | Calenduloside H | IUPAC name | 6-[[4,4,6a,6b,11,11,14b-heptamethyl-8a-[3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl]oxycarbonyl-1,2,3,4a,5,6,7,8,9,10,12,12a,14,14a-tetradecahydropicen-3-yl]oxy]-3,5-dihydroxy-4-[3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl]oxyoxane-2-carboxylic acid |
| Molecular formula | C48H76O19 |
Experimental data
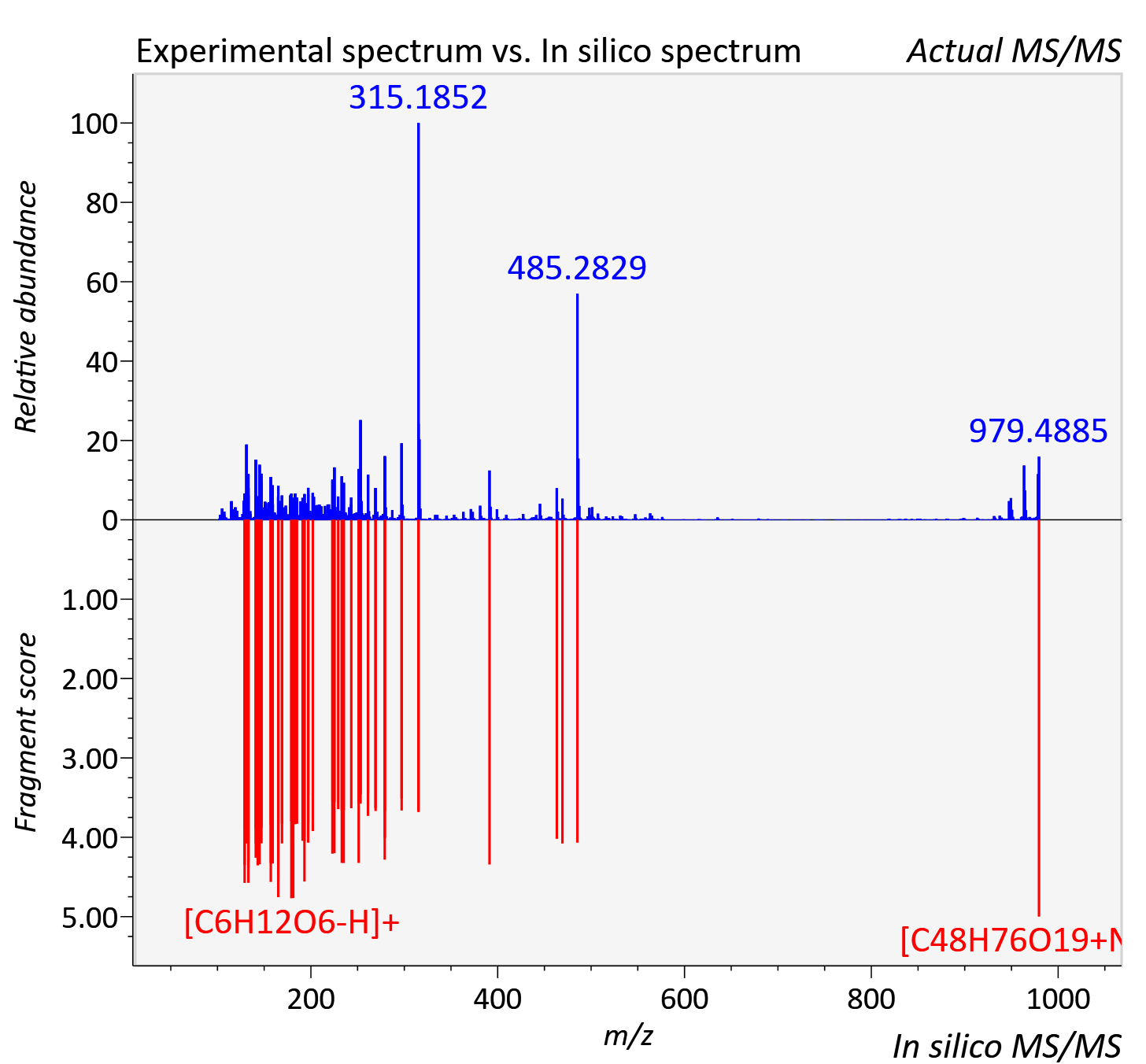
| Retention time | 7.12 |
|---|---|
| Adduct | [M+Na]+ |
| Actual mz | 979.487 | Theoretical mz | 979.487 |
| Error | 0.08 |
| Ionizaton mode | Positive |
| Instrument type | LC-MS/MS-QTOF, spectrum predicted by MS-DIAL v4.9 integrated with MS-FINDER 3.60 |
| Score | 6.9935 |
Identifiers and class information
| Inchi key | QZMAEZWZCGBZFK-UHFFFAOYNA-N |
|---|---|
| Smiles | O=C(O)C1OC(OC2CCC3(C)C4CC=C5C6CC(C)(C)CCC6(C(=O)OC7OC(CO)C(O)C(O)C7O)CCC5(C)C4(C)CCC3C2(C)C)C(O)C(OC8OC(CO)C(O)C(O)C8O)C1O |
| Superclass | Lipids and lipid-like molecules |
| Class | Prenol lipids |
Plant source
Pharmacokinetic properties
Compound-target network
Protein targets associated with phytocompound
| Uniprot ID | Gene name | Target name | TTD_ID | Prediction source |
|---|---|---|---|---|
| P29350 | PTPN6 | Protein-tyrosine phosphatase 1C | T98264 | SEA |
| P18031 | PTPN1 | Protein-tyrosine phosphatase 1B | T16347 | SEA |
| P17706 | PTPN2 | T-cell protein-tyrosine phosphatase | T49156 | SEA |
| P06746 | POLB | DNA polymerase beta (by homology) | T06958 | SEA |
| P13726 | F3 | Coagulation factor VII/tissue factor | T72702 | SEA |
| P15692 | VEGFA | Vascular endothelial growth factor A | T20761 | SEA |
| P05230 | FGF1 | Acidic fibroblast growth factor | T18639 | SEA |
| P09038 | FGF2 | Basic fibroblast growth factor | T31621 | SEA |
| P60568 | IL2 | Interleukin-2 | T61698 | SEA |
| P16885 | PLCG2 | Phospholipase C-gamma-2 | T93922 | SEA |
Target associated diseases
| TTD_ID | Disease_ID | Disease name | ICD_11 | Uniprot ID | Gene names |
|---|---|---|---|---|---|
| T16347 | DI0009 | Acute diabete complication | [ICD-11: 5A2Y] | P18031 | PTPN1 |
| T16347 | DI0062 | Breast cancer | [ICD-11: 2C60-2C6Y] | P18031 | PTPN1 |
| T16347 | DI0308 | Obesity | [ICD-11: 5B80-5B81] | P18031 | PTPN1 |
| T16347 | DI0417 | Type 2 diabetes mellitus | [ICD-11: 5A11] | P18031 | PTPN1 |
| T49156 | DI0391 | Solid tumour/cancer | [ICD-11: 2A00-2F9Z] | P17706 | PTPN2 |
| T06958 | DI0391 | Solid tumour/cancer | [ICD-11: 2A00-2F9Z] | P06746 | POLB |
| T72702 | DI0075 | Cervical cancer | [ICD-11: 2C77] | P13726 | F3 |
| T20761 | DI0095 | Colorectal cancer | [ICD-11: 2B91] | P15692 | VEGFA |
| T20761 | DI0365 | Retinopathy | [ICD-11: 9B71] | P15692 | VEGFA |
| T20761 | DI0430 | Vascular system developmental anomaly | [ICD-11: LA90] | P15692 | VEGFA |
| T18639 | DI0081 | Chronic arterial occlusive disease | [ICD-11: BD4Z] | P05230 | FGF1 |
| T18639 | DI0102 | Coronary atherosclerosis | [ICD-11: BA52] | P05230 | FGF1 |
| T31621 | DI0005 | Acne vulgaris | [ICD-11: ED80] | P09038 | FGF2 |
| T61698 | DI0275 | Multiple sclerosis | [ICD-11: 8A40] | P60568 | IL2 |
| T61698 | DI0361 | Renal cell carcinoma | [ICD-11: 2C90] | P60568 | IL2 |
