Compound details
Sanggenon E
| Compound ID | CDAMM02025 |
|---|---|
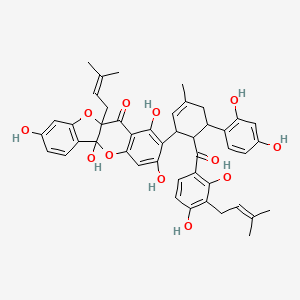
| Common name | Sanggenon E | IUPAC name | 2-[6-[2,4-dihydroxy-3-(3-methylbut-2-enyl)benzoyl]-5-(2,4-dihydroxyphenyl)-3-methylcyclohex-2-en-1-yl]-1,3,5a,8-tetrahydroxy-10a-(3-methylbut-2-enyl)-[1]benzofuro[3,2-b]chromen-11-one |
| Molecular formula | C45H44O12 |
Experimental data
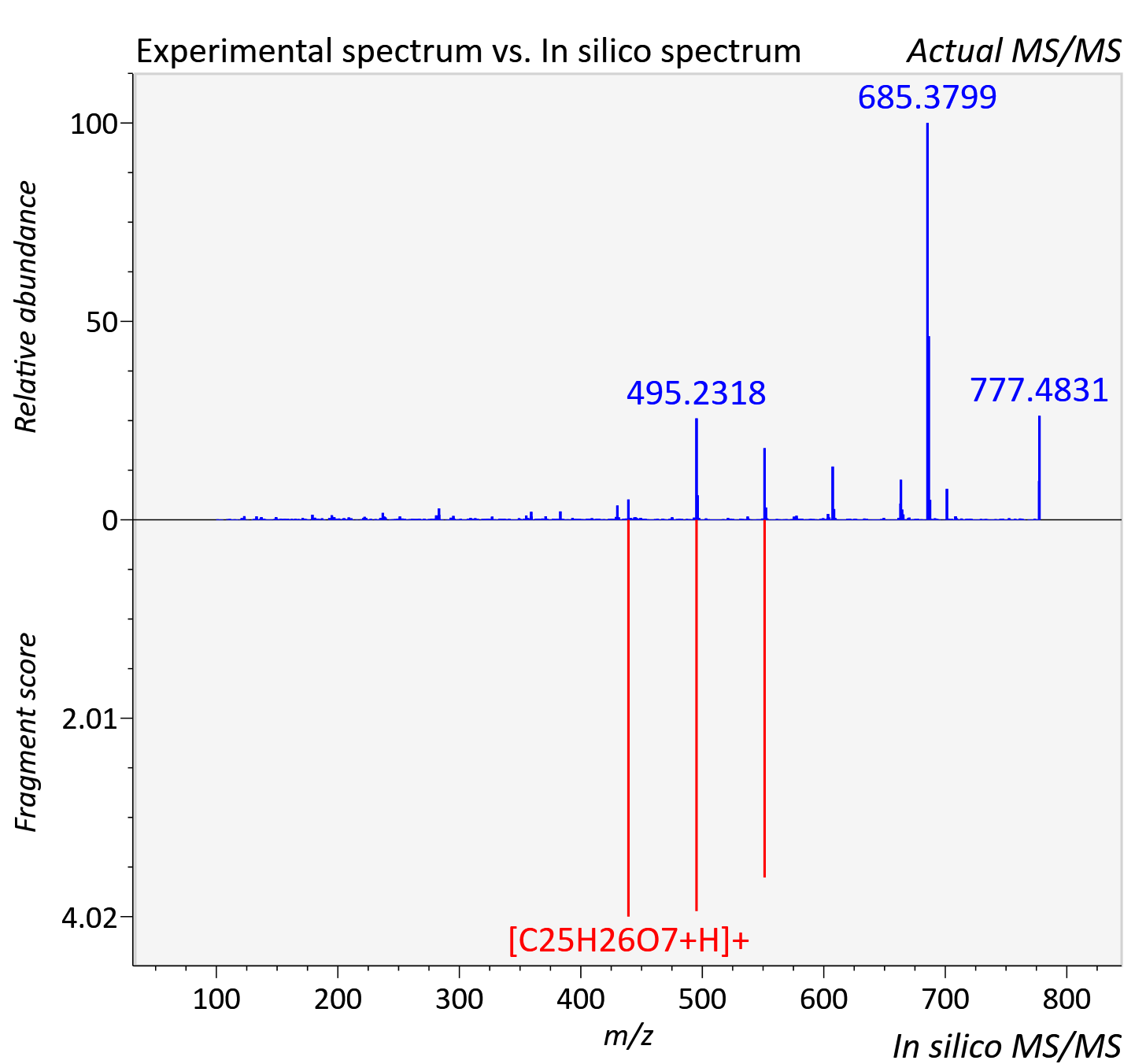
| Retention time | 19.42 |
|---|---|
| Adduct | [M+H]+ |
| Actual mz | 777.29 | Theoretical mz | 777.29 |
| Error | 0.23 |
| Ionizaton mode | Positive |
| Instrument type | LC-MS/MS-QTOF, spectrum predicted by MS-DIAL v4.9 integrated with MS-FINDER 3.60 |
| Score | 6.2757 |
Identifiers and class information
| Inchi key | CUJJTBMGUHNKPO-GXQCSOQPNA-N |
|---|---|
| Smiles | O=C(C1=CC=C(O)C(=C1O)CC=C(C)C)C2C(C=C(C)CC2C3=CC=C(O)C=C3O)C4=C(O)C=C5OC6(O)C7=CC=C(O)C=C7OC6(C(=O)C5=C4O)CC=C(C)C |
| Superclass | Phenylpropanoids and polyketides |
| Class | Diarylheptanoids |
Plant source
Pharmacokinetic properties
Target associated diseases
| TTD_ID | Disease_ID | Disease name | ICD_11 | Uniprot ID | Gene names |
|---|---|---|---|---|---|
| T16347 | DI0009 | Acute diabete complication | [ICD-11: 5A2Y] | P18031 | PTPN1 |
| T16347 | DI0062 | Breast cancer | [ICD-11: 2C60-2C6Y] | P18031 | PTPN1 |
| T16347 | DI0308 | Obesity | [ICD-11: 5B80-5B81] | P18031 | PTPN1 |
| T16347 | DI0417 | Type 2 diabetes mellitus | [ICD-11: 5A11] | P18031 | PTPN1 |
| T97035 | DI0007 | Acquired hypermelanosis | [ICD-11: ED60] | P14679 | TYR |
| T97035 | DI0008 | Acquired hypomelanotic disorder | [ICD-11: ED63] | P14679 | TYR |
