Compound details
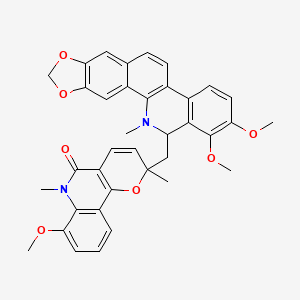
Simulanoquinoline
| Compound ID | CDAMM01475 |
|---|---|
| Common name | Simulanoquinoline | IUPAC name | 2-[(1,2-dimethoxy-12-methyl-13H-[1,3]benzodioxolo[5,6-c]phenanthridin-13-yl)methyl]-7-methoxy-2,6-dimethylpyrano[3,2-c]quinolin-5-one |
| Molecular formula | C37H34N2O7 |
Experimental data
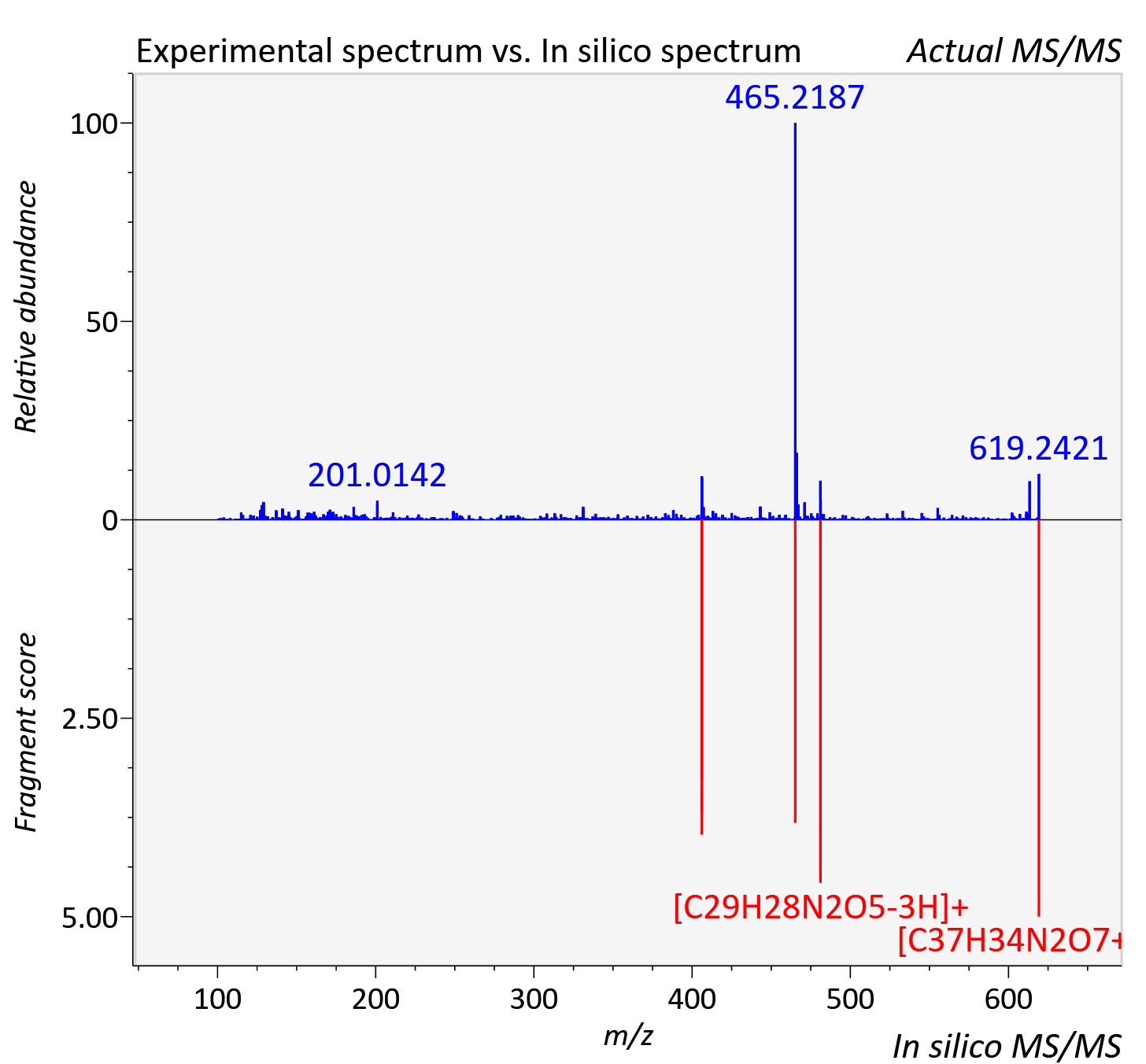
| Retention time | 3.49 |
|---|---|
| Adduct | [M+H]+ |
| Actual mz | 619.244 | Theoretical mz | 619.244 |
| Error | 0.04 |
| Ionizaton mode | Positive |
| Instrument type | LC-MS/MS-QTOF, spectrum predicted by MS-DIAL v4.9 integrated with MS-FINDER 3.60 |
| Score | 7.6093 |
Identifiers and class information
| Inchi key | NUVWARFQHKLGOS-UHFFFAOYNA-N |
|---|---|
| Smiles | O=C1C=2C=CC(OC2C=3C=CC=C(OC)C3N1C)(C)CC4C=5C(OC)=C(OC)C=CC5C6=CC=C7C=C8OCOC8=CC7=C6N4C |
| Superclass | Alkaloids and derivatives |
| Class | Benzophenanthridine alkaloids |
Plant source
Pharmacokinetic properties
Compound-target network
Target associated diseases
| TTD_ID | Disease_ID | Disease name | ICD_11 | Uniprot ID | Gene names |
|---|---|---|---|---|---|
| T60366 | DI0020 | African trypanosomiasis | [ICD-11: 1F51] | P11926 | ODC1 |
| T81443 | DI0022 | Allergic/hypersensitivity disorder | [ICD-11: 4A80-4A8Z] | O00206 | TLR4 |
| T81443 | DI0346 | Prostate cancer | [ICD-11: 2C82] | O00206 | TLR4 |
| T81443 | DI0375 | Sepsis | [ICD-11: 1G40-1G41] | O00206 | TLR4 |
| T37510 | DI0073 | Cerebral ischaemia | [ICD-11: 8B1Z] | P12259 | F5 |
| T37510 | DI0375 | Sepsis | [ICD-11: 1G40-1G41] | P12259 | F5 |
| T85733 | DI0062 | Breast cancer | [ICD-11: 2C60-2C6Y] | Q02880 | TOP2B |
| T85733 | DI0076 | Chemoprotection | [ICD-11: N.A.] | Q02880 | TOP2B |
