Compound details
Cyclovirobuxeine A
| Compound ID | CDAMM01422 |
|---|---|
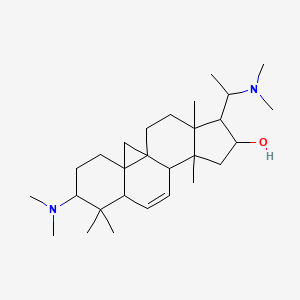
| Common name | Cyclovirobuxeine A | IUPAC name | 6-(dimethylamino)-15-[1-(dimethylamino)ethyl]-7,7,12,16-tetramethylpentacyclo[9.7.0.01,3.03,8.012,16]octadec-9-en-14-ol |
| Molecular formula | C28H48N2O |
Experimental data
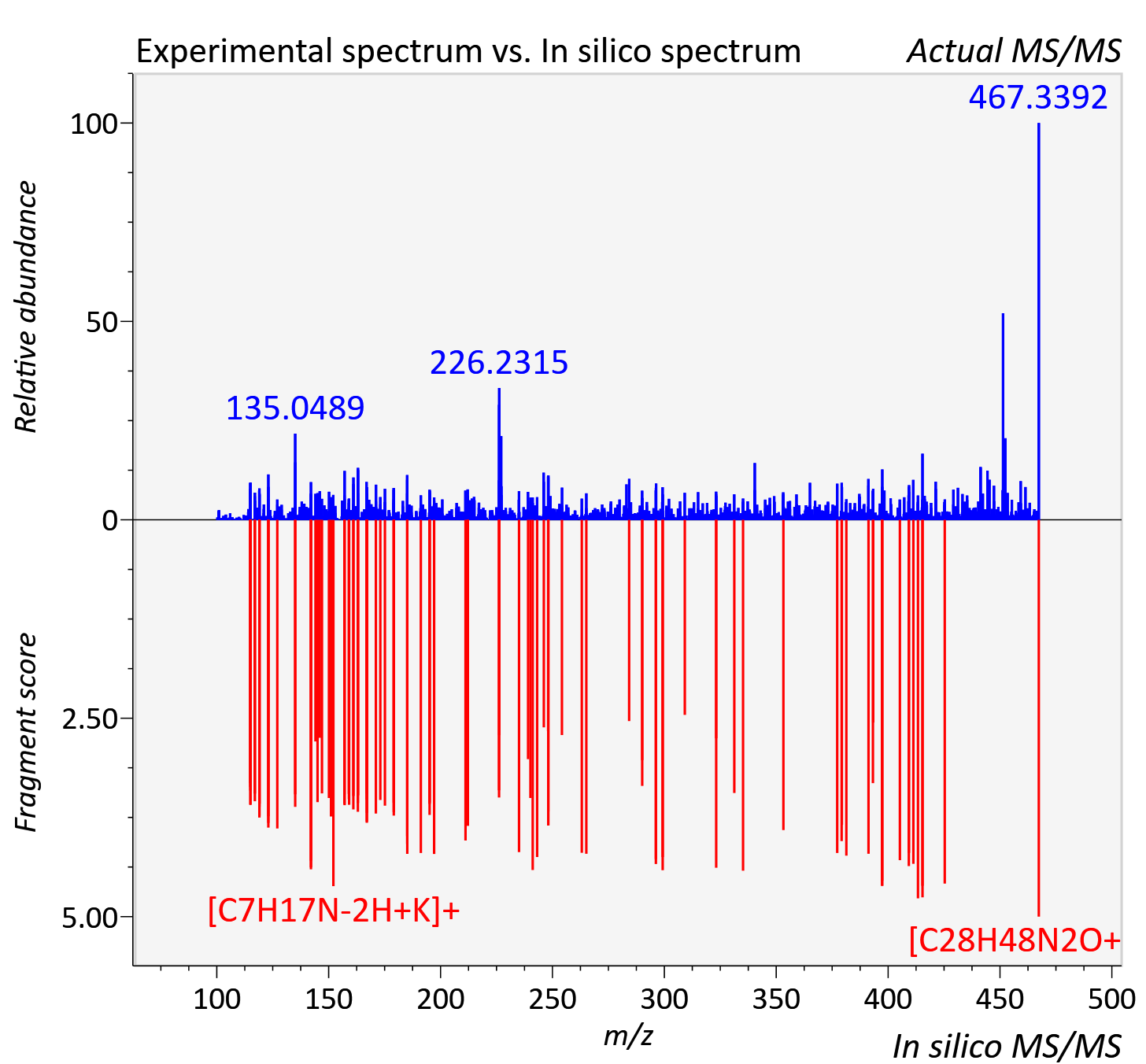
| Retention time | 14.94 |
|---|---|
| Adduct | [M+K]+ |
| Actual mz | 467.343 | Theoretical mz | 467.34 |
| Error | 5.23 |
| Ionizaton mode | Positive |
| Instrument type | LC-MS/MS-QTOF, spectrum predicted by MS-DIAL v4.9 integrated with MS-FINDER 3.60 |
| Score | 6.1856 |
Identifiers and class information
| Inchi key | UVWOWJBPZRXVCO-XNIUDBTQNA-N |
|---|---|
| Smiles | OC1CC2(C)C3C=CC4C(C)(C)C(N(C)C)CCC54CC35CCC2(C)C1C(N(C)C)C |
| Superclass | Lipids and lipid-like molecules |
| Class | Prenol lipids |
Plant source
Pharmacokinetic properties
Compound-target network
Protein targets associated with phytocompound
| Uniprot ID | Gene name | Target name | TTD_ID | Prediction source |
|---|---|---|---|---|
| Q99720 | SIGMAR1 | Sigma opioid receptor | T46360 | SwissTargetPrediction |
| P48449 | LSS | Lanosterol synthase | T56175 | SwissTargetPrediction |
| P11511 | CYP19A1 | Cytochrome P450 19A1 | T13260 | SwissTargetPrediction |
| Q15465 | SHH | Sonic hedgehog protein (by homology) | T12075 | SwissTargetPrediction |
| P41145 | OPRK1 | Kappa Opioid receptor | T60693 | SwissTargetPrediction |
| P32245 | MC4R | Melanocortin receptor 4 | T72458 | SwissTargetPrediction |
| P41968 | MC3R | Melanocortin receptor 3 | T76846 | SwissTargetPrediction |
| P05093 | CYP17A1 | Cytochrome P450 17A1 | T89041 | SwissTargetPrediction |
| P32246 | CCR1 | C-C chemokine receptor type 1 | T16016 | SwissTargetPrediction |
| P41146 | OPRL1 | Nociceptin receptor | T52921 | SwissTargetPrediction |
| P24557 | TBXAS1 | Thromboxane-A synthase | T78356 | SwissTargetPrediction |
| P33032 | MC5R | Melanocortin receptor 5 | T95302 | SwissTargetPrediction |
Target associated diseases
| TTD_ID | Disease_ID | Disease name | ICD_11 | Uniprot ID | Gene names |
|---|---|---|---|---|---|
| T46360 | DI0105 | Cough | [ICD-11: MD12] | Q99720 | SIGMAR1 |
| T56175 | DI0035 | Arterial occlusive disease | [ICD-11: BD40] | P48449 | LSS |
| T13260 | DI0062 | Breast cancer | [ICD-11: 2C60-2C6Y] | P11511 | CYP19A1 |
| T13260 | DI0108 | Cushing syndrome | [ICD-11: 5A70] | P11511 | CYP19A1 |
| T60693 | DI0304 | Non-specific cutaneous vascular symptom | [ICD-11: ME64] | P41145 | OPRK1 |
| T60693 | DI0324 | Pain | [ICD-11: MG30-MG3Z] | P41145 | OPRK1 |
| T60693 | DI0349 | Pruritus | [ICD-11: EC90] | P41145 | OPRK1 |
| T72458 | DI0192 | Hypoactive sexual desire dysfunction | [ICD-11: HA00] | P32245 | MC4R |
| T72458 | DI0228 | Large intestine motility disorder | [ICD-11: DB32] | P32245 | MC4R |
| T72458 | DI0308 | Obesity | [ICD-11: 5B80-5B81] | P32245 | MC4R |
| T76846 | DI0308 | Obesity | [ICD-11: 5B80-5B81] | P41968 | MC3R |
| T76846 | DI0378 | Sexual dysfunction | [ICD-11: HA00-HA01] | P41968 | MC3R |
| T76846 | DI0417 | Type 2 diabetes mellitus | [ICD-11: 5A11] | P41968 | MC3R |
| T89041 | DI0346 | Prostate cancer | [ICD-11: 2C82] | P05093 | CYP17A1 |
| T16016 | DI0120 | Diabetes mellitus | [ICD-11: 5A10] | P32246 | CCR1 |
| T16016 | DI0366 | Rheumatoid arthritis | [ICD-11: FA20] | P32246 | CCR1 |
| T52921 | DI0039 | Atopic eczema | [ICD-11: EA80] | P41146 | OPRL1 |
| T52921 | DI0117 | Depression | [ICD-11: 6A70-6A7Z] | P41146 | OPRL1 |
| T52921 | DI0173 | Headache | [ICD-11: 8A80-8A84] | P41146 | OPRL1 |
| T52921 | DI0175 | Heart failure | [ICD-11: BD10-BD1Z] | P41146 | OPRL1 |
| T78356 | DI0030 | Angina pectoris | [ICD-11: BA40] | P24557 | TBXAS1 |
| T95302 | DI0005 | Acne vulgaris | [ICD-11: ED80] | P33032 | MC5R |
| T95302 | DI0371 | Sebaceous gland disorder | [ICD-11: ED91] | P33032 | MC5R |
