Compound details
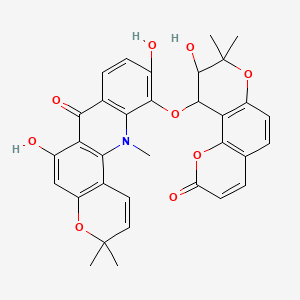
Neoacrimarine C
| Compound ID | CDAMM01343 |
|---|---|
| Common name | Neoacrimarine C | IUPAC name | 6,10-dihydroxy-11-[(9-hydroxy-8,8-dimethyl-2-oxo-9,10-dihydropyrano[2,3-f]chromen-10-yl)oxy]-3,3,12-trimethylpyrano[2,3-c]acridin-7-one |
| Molecular formula | C33H29NO9 |
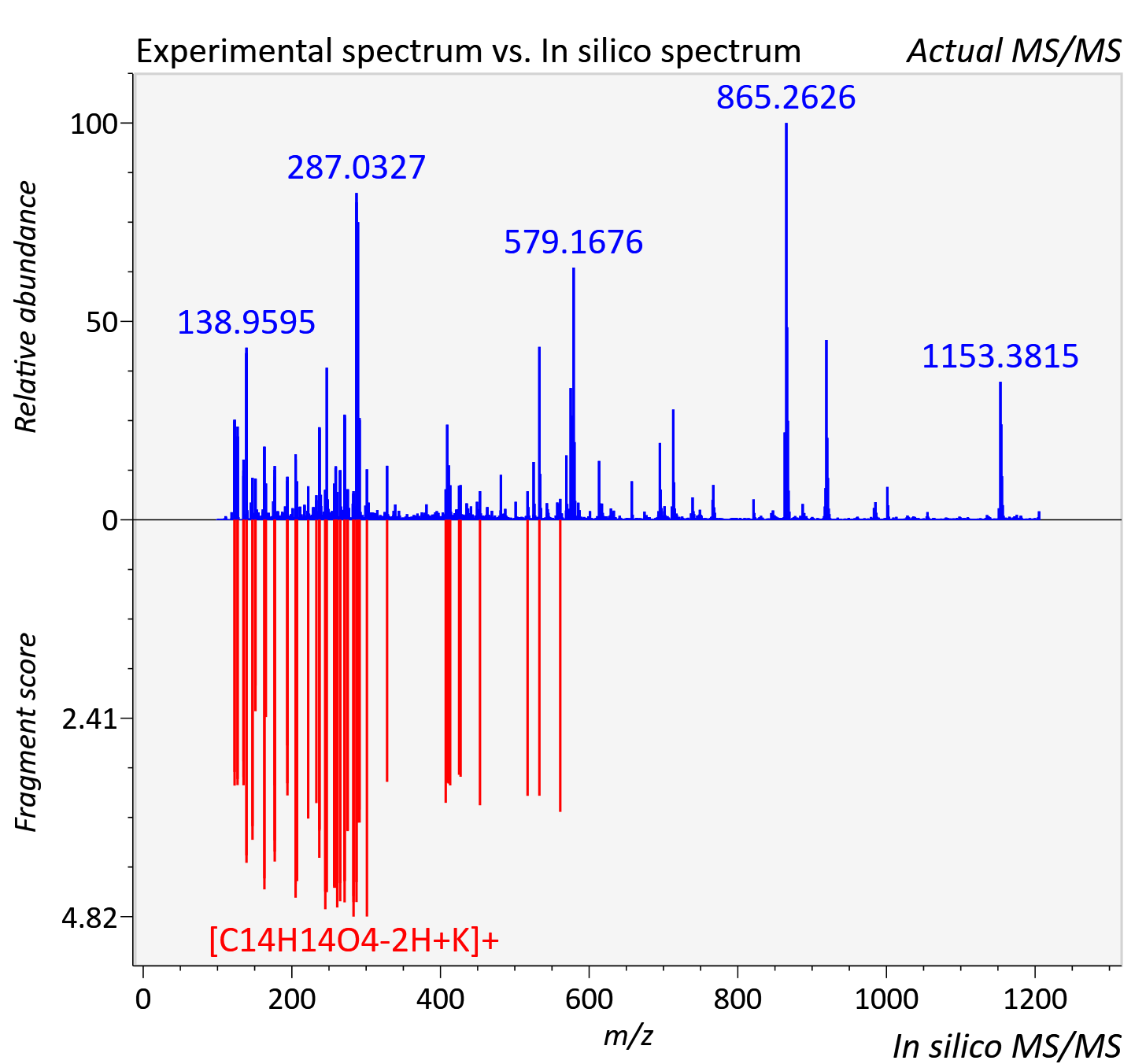
Experimental data
| Retention time | 0.26 |
|---|---|
| Adduct | [2M+K]+ |
| Actual mz | 1205.33 | Theoretical mz | 1205.33 |
| Error | 4.28 |
| Ionizaton mode | Positive |
| Instrument type | LC-MS/MS-QTOF, spectrum predicted by MS-DIAL v4.9 integrated with MS-FINDER 3.60 |
| Score | 6.4135 |
Identifiers and class information
| Inchi key | NQAQWRPJTRYUKE-RFTMCNCJNA-N |
|---|---|
| Smiles | O=C1OC2=C(C=C1)C=CC=3OC(C)(C)C(O)C(OC=4C(O)=CC=C5C(=O)C6=C(O)C=C7OC(C=CC7=C6N(C45)C)(C)C)C32 |
| Superclass | Organoheterocyclic compounds |
| Class | Quinolines and derivatives |
