Compound details
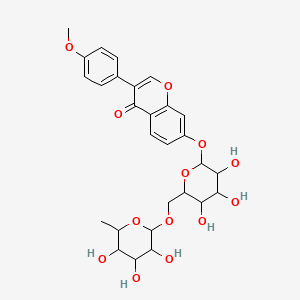
Formononetin 7-O-rutinoside
| Compound ID | CDAMM01310 |
|---|---|
| Common name | Formononetin 7-O-rutinoside | IUPAC name | 3-(4-methoxyphenyl)-7-[3,4,5-trihydroxy-6-[(3,4,5-trihydroxy-6-methyloxan-2-yl)oxymethyl]oxan-2-yl]oxychromen-4-one |
| Molecular formula | C28H32O13 |
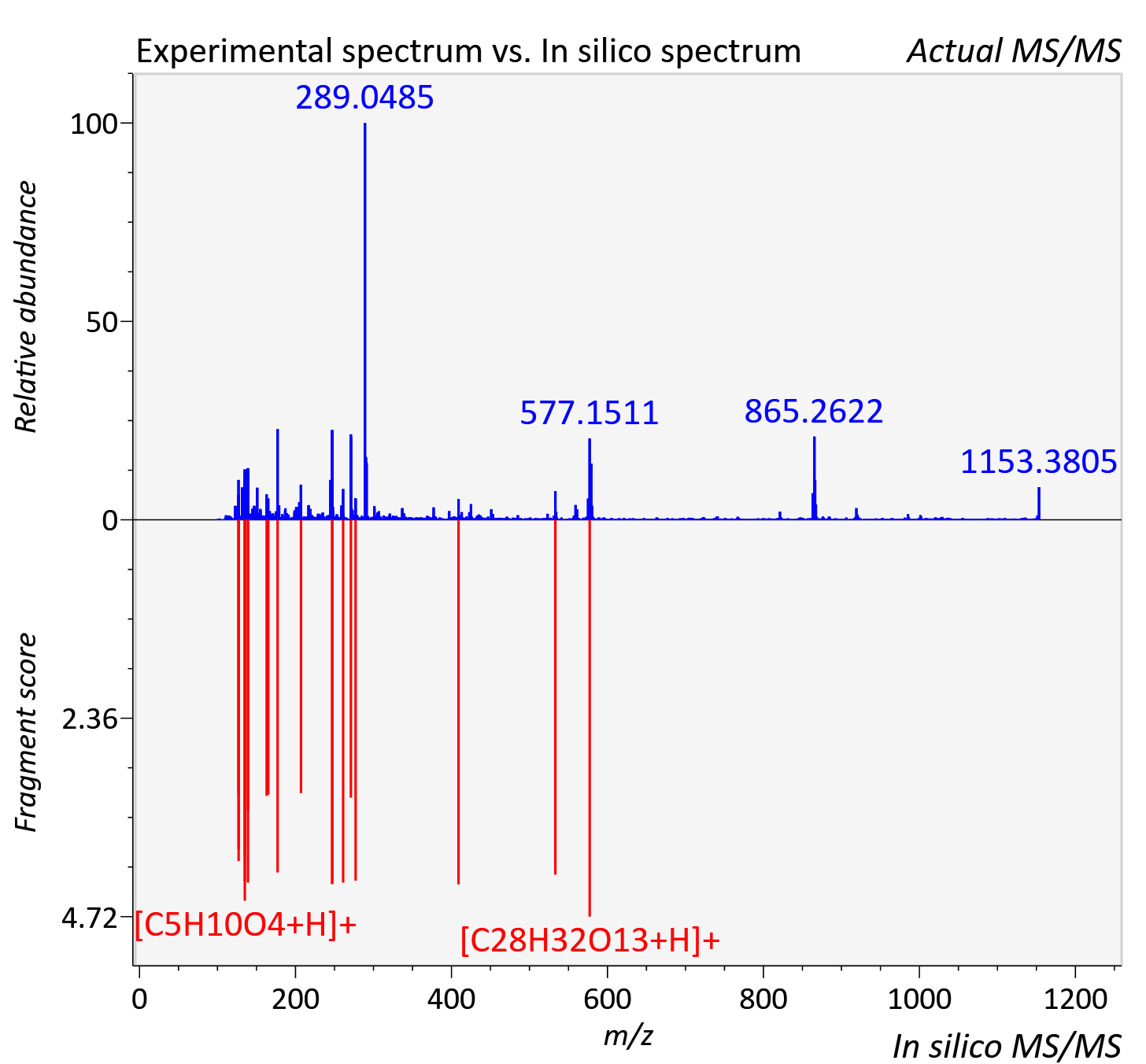
Experimental data
| Retention time | 0.47 |
|---|---|
| Adduct | [2M+H]+ |
| Actual mz | 1153.38 | Theoretical mz | 1153.38 |
| Error | 2.36 |
| Ionizaton mode | Positive |
| Instrument type | LC-MS/MS-QTOF, spectrum predicted by MS-DIAL v4.9 integrated with MS-FINDER 3.60 |
| Score | 6.9183 |
Identifiers and class information
| Inchi key | ZSCRYAYQFLBRDF-OEPCLMMTNA-N |
|---|---|
| Smiles | O=C1C(=COC2=CC(OC3OC(COC4OC(C)C(O)C(O)C4O)C(O)C(O)C3O)=CC=C12)C=5C=CC(OC)=CC5 |
| Superclass | Phenylpropanoids and polyketides |
| Class | Isoflavonoids |
Pharmacokinetic properties
Compound-target network
Protein targets associated with phytocompound
| Uniprot ID | Gene name | Target name | TTD_ID | Prediction source |
|---|---|---|---|---|
| O43570 | CA12 | Carbonic anhydrase XII | T16987 | SEA |
| P43166 | CA7 | Carbonic anhydrase VII | T37541 | SEA |
| P22748 | CA4 | Carbonic anhydrase IV | T53378 | SEA |
| P01375 | TNF | TNF-alpha | T20178 | SwissTargetPrediction |
| P60568 | IL2 | Interleukin-2 | T61698 | SwissTargetPrediction and SEA |
| P30837 | ALDH1B1 | Acetaldehyde dehydrogenase | T99641 | SwissTargetPrediction and SEA |
| P04746 | AMY2A | Pancreatic alpha-amylase | T86918 | SEA |
| P14679 | TYR | Tyrosinase | T97035 | SEA |
| Q9GZQ4 | NMUR2 | Neuromedin-U receptor 2 | T04210 | SEA |
| Q16678 | CYP1B1 | Cytochrome P450 1B1 | T92521 | SEA |
| P16152 | CBR1 | Carbonyl reductase [NADPH] 1 | T70518 | SEA |
| Q9NZ08 | ERAP1 | Endoplasmic reticulum aminopeptidase 1 | T72849 | SEA |
| Q15391 | P2RY14 | Purinergic receptor P2Y14 | T16555 | SEA |
| P49862 | KLK7 | Kallikrein-7 | T79155 | SEA |
| P30837 | ALDH1B1 | Acetaldehyde dehydrogenase | T99641 | SEA |
Target associated diseases
| TTD_ID | Disease_ID | Disease name | ICD_11 | Uniprot ID | Gene names |
|---|---|---|---|---|---|
| T16987 | DI0046 | Bacterial infection | [ICD-11: 1A00-1C4Z] | O43570 | CA12 |
| T16987 | DI0372 | Seborrhoeic dermatitis | [ICD-11: EA81] | O43570 | CA12 |
| T53378 | DI0046 | Bacterial infection | [ICD-11: 1A00-1C4Z] | P22748 | CA4 |
| T53378 | DI0166 | Glaucoma | [ICD-11: 9C61] | P22748 | CA4 |
| T53378 | DI0372 | Seborrhoeic dermatitis | [ICD-11: EA81] | P22748 | CA4 |
| T20178 | DI0035 | Arterial occlusive disease | [ICD-11: BD40] | P01375 | TNF |
| T20178 | DI0274 | Multiple myeloma | [ICD-11: 2A83] | P01375 | TNF |
| T20178 | DI0351 | Psoriasis | [ICD-11: EA90] | P01375 | TNF |
| T20178 | DI0366 | Rheumatoid arthritis | [ICD-11: FA20] | P01375 | TNF |
| T61698 | DI0275 | Multiple sclerosis | [ICD-11: 8A40] | P60568 | IL2 |
| T61698 | DI0361 | Renal cell carcinoma | [ICD-11: 2C90] | P60568 | IL2 |
| T99641 | DI0396 | Substance abuse | [ICD-11: 6C40] | P30837 | ALDH1B1 |
| T86918 | DI0110 | Cystic fibrosis | [ICD-11: CA25] | P04746 | AMY2A |
| T86918 | DI0328 | Pancreatic malfunction | [ICD-11: DC30-DC3Z] | P04746 | AMY2A |
| T97035 | DI0007 | Acquired hypermelanosis | [ICD-11: ED60] | P14679 | TYR |
| T97035 | DI0008 | Acquired hypomelanotic disorder | [ICD-11: ED63] | P14679 | TYR |
| T70518 | DI0037 | Asthma | [ICD-11: CA23] | P16152 | CBR1 |
| T99641 | DI0396 | Substance abuse | [ICD-11: 6C40] | P30837 | ALDH1B1 |
