Compound details
N-{3-[2-(methylsulfanyl)ethyl]-2,5-dioxo-2,3,4,5-tetrahydro-1H-1,4-benzodiazepin-7-yl}-4-(4-oxo-3,4-dihydro-1,2,3-benzotriazin-3-yl)butanamide
| Compound ID | CDAMM00195 |
|---|---|
| Common name | N-{3-[2-(methylsulfanyl)ethyl]-2,5-dioxo-2,3,4,5-tetrahydro-1H-1,4-benzodiazepin-7-yl}-4-(4-oxo-3,4-dihydro-1,2,3-benzotriazin-3-yl)butanamide | IUPAC name | N-[3-(2-methylsulfanylethyl)-2,5-dioxo-3,4-dihydro-1H-1,4-benzodiazepin-7-yl]-4-(4-oxo-1,2,3-benzotriazin-3-yl)butanamide |
| Molecular formula | C23H24N6O4S |
Experimental data
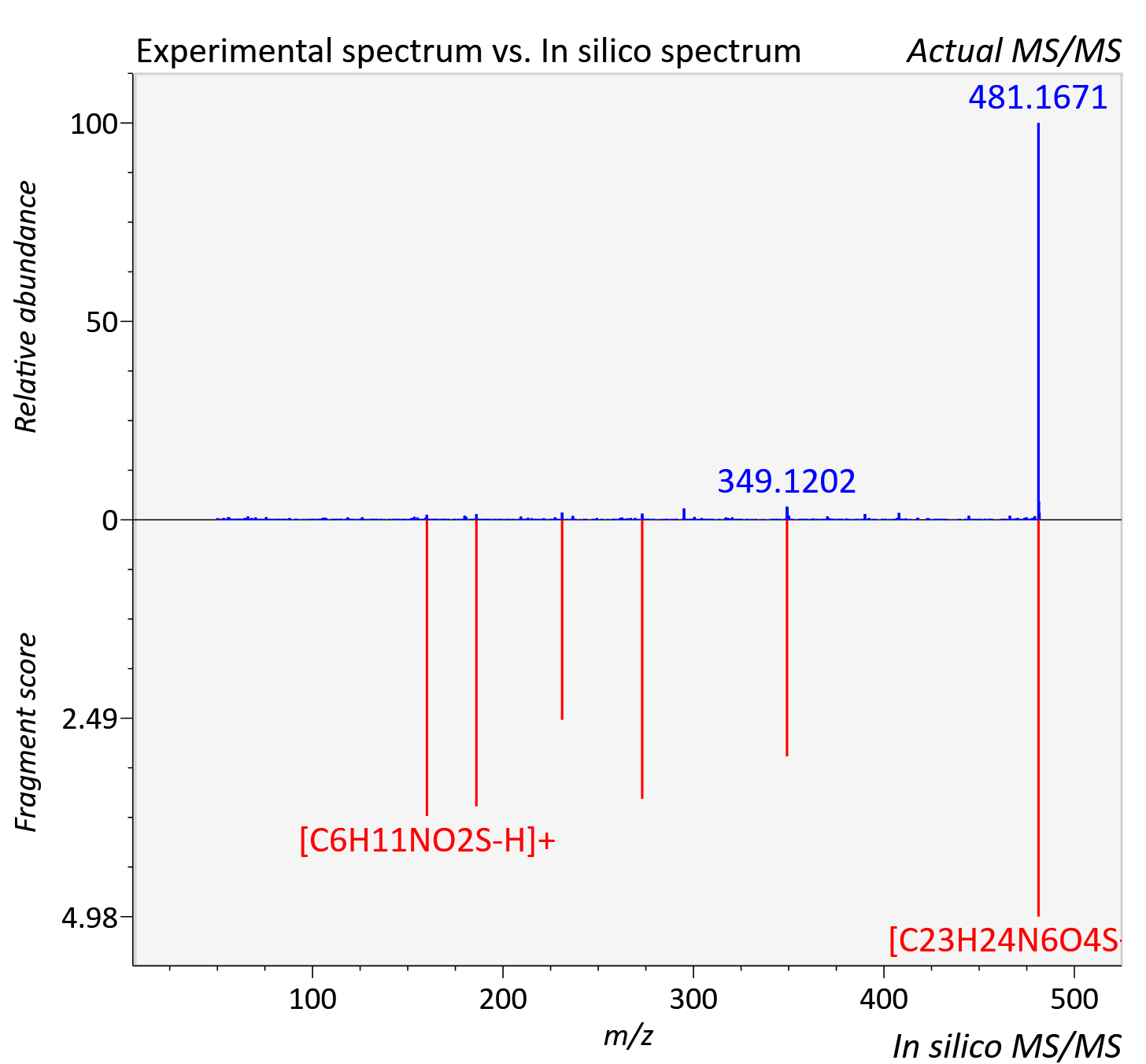
| Retention time | 47.97 |
|---|---|
| Adduct | [M+H]+ |
| Actual mz | 481.166 | Theoretical mz | 481.165 |
| Error | 1.76 |
| Ionizaton mode | Positive |
| Instrument type | LC-MS/MS-QTOF, spectrum predicted by MS-DIAL v4.9 integrated with MS-FINDER 3.60 |
| Score | 5.2483 |
Identifiers and class information
| Inchi key | NGOYNQGGWBCEAM-UHFFFAOYSA-N |
|---|---|
| Smiles | CSCCC1C(=O)NC2=C(C=C(C=C2)NC(=O)CCCN3C(=O)C4=CC=CC=C4N=N3)C(=O)N1 |
| Superclass | Organoheterocyclic compounds |
| Class | Benzodiazepines |
Plant source
Pharmacokinetic properties
Compound-target network
Protein targets associated with phytocompound
| Uniprot ID | Gene name | Target name | TTD_ID | Prediction source |
|---|---|---|---|---|
| P08575 | PTPRC | Leukocyte common antigen | T43115 | SEA |
| P42575 | CASP2 | Caspase-2 | T09128 | SEA |
| O14746 | TERT | Telomerase reverse transcriptase | T86052 | SEA |
| P17301 | ITGA2 | Integrin alpha-2 | T55293 | SEA |
| P01130 | LDLR | LDL receptor | T94692 | SEA |
| Q9NYN9 | PTPN13 | FAP-1 messenger RNA | T09672 | SEA |
| Q4QXX7 | MRGPRX2 | Mas-related gene 2 | T38187 | SEA |
Target associated diseases
| TTD_ID | Disease_ID | Disease name | ICD_11 | Uniprot ID | Gene names |
|---|---|---|---|---|---|
| T43115 | DI0012 | Acute myeloid leukaemia | [ICD-11: 2A60] | P08575 | PTPRC |
| T43115 | DI0414 | Transplanted organ/tissue | [ICD-11: QB63] | P08575 | PTPRC |
| T86052 | DI0184 | Human immunodeficiency virus disease | [ICD-11: 1C60-1C62] | O14746 | TERT |
| T86052 | DI0284 | Myelodysplastic syndrome | [ICD-11: 2A37] | O14746 | TERT |
| T86052 | DI0326 | Pancreatic cancer | [ICD-11: 2C10] | O14746 | TERT |
| T55293 | DI0095 | Colorectal cancer | [ICD-11: 2B91] | P17301 | ITGA2 |
| T55293 | DI0391 | Solid tumour/cancer | [ICD-11: 2A00-2F9Z] | P17301 | ITGA2 |
| T94692 | DI0238 | Lung cancer | [ICD-11: 2C25] | P01130 | LDLR |
